An established need
At Harvard Medical School, Allen “Jack” Latham recognizes the need for improved blood processing systems.
At Harvard Medical School, Allen “Jack” Latham recognizes the need for improved blood processing systems.
Building on the work of Dr. Edwin Cohn, Jack Latham invents a transparent, disposable centrifuge bowl, known as the Latham bowl.
Jack Latham founds Haemonetics in Natick, MA, dedicated to improving the safety and quality of blood supply.
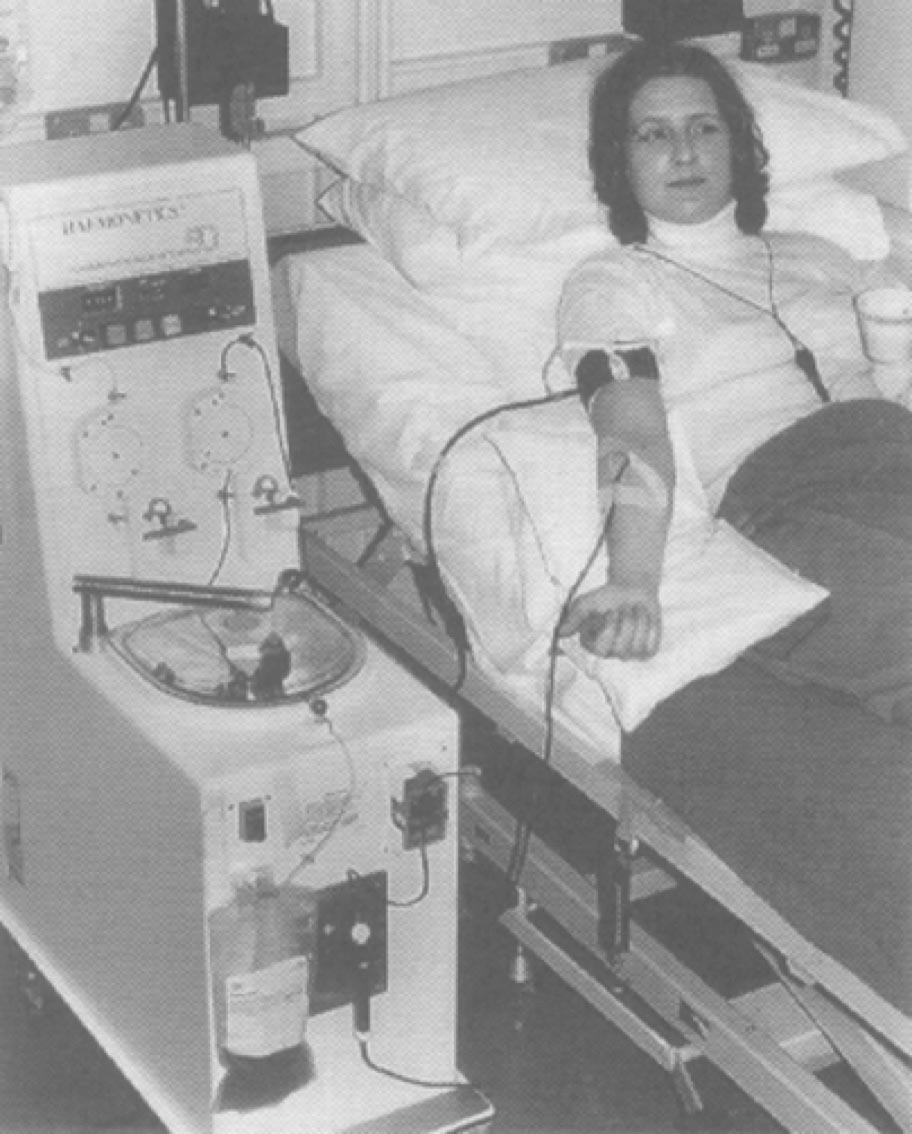
The Model 30 system is introduced, marking the beginning of the modern age of apheresis.
The Cell Saver® system is introduced as the first surgical washed blood salvage technology, soon to become the standard of care for hospitals.
With new developments like Model 115 and Model 30-S, the company continues to grow, opening its Braintree, MA, facility at 400 Wood Road.
The company successfully completes an over-the-counter initial public offering, enabling future growth.
Cell Saver® 3 launches as the first system to automate surgical blood salvage.
The company opens its first international headquarters in Nyon, Switzerland.
Haemonetics launches the PCS® plasma collection system, significantly cutting the time needed to collect plasma and increasing donor safety.
Cell Saver® 4, the first automated high-speed blood salvage technology, is launched.
A group of private investors, including present and former employees purchases the company.
The Department of Defense contracts for deployment of the Cell Saver® 4 and Model 115 systems at military installations and on ships worldwide.
Jack Latham is named “New England Inventor of the Year” by the Boston Patent Law Association.
The facility in Leetsdale, PA, opens as a distribution center.
The company is taken public again, successfully completing an initial public offering (HAE) on the New York Stock Exchange.
The company begins direct sales and service in Japan.
The company develops the MCS® mobile apheresis system, making it possible to provide blood component therapy to patients at blood banks.
The PCS® 2 plasma collection system launches.
The blow-molded version of the plasma collection bowl is introduced.
Manufacturing of the blow-molded bowl begins at the Leetsdale, PA, facility, the first time a blow-molded bowl is manufactured at a Haemonetics facility.
The MCS®+ mobile platelet collection system launches.
The company acquires the Intravenous Solutions manufacturing facility in Union, SC
The company acquires a plasma collection bottle manufacturing facility and Fifth Dimension Information Systems.
The company reaches the $1 billion market cap.
The American Red Cross launches a double red cell collection program using the MCS®+ 8150 system.
The ACP® 215 system cell washing protocol is introduced.
The company launches a proprietary plasma collection bowl that removes white blood cells from plasma without requiring an attached filtration device.
The cardioPAT® surgical blood salvage system and the SmartSuction Harmony® surgical suction system are introduced.
The company acquires Haemoscope and the TEG® 5000 hemostasis analyzer system.
The company acquires Altivation Software and Neoteric Technology, both information management companies.
The company acquires Global Med Technologies, Inc., a California-based medical information technology company.
Cell Saver® Elite® autotransfusion system launches.
NexGen DMS™ donor management software launches.
The company acquires the blood collection, filtration, and processing product lines from Pall Corporation.
The BloodTrack® HaemoBank® blood management solution is introduced.
The TEG® 6s hemostasis analyzer and TEG Manager® software launch.
The Penang, Malaysia, manufacturing site holds its grand opening, while the Tijuana, Mexico, manufacturing facility expands.
Cell Saver® Elite®+ autologous blood recovery system launches with enhancements, including new design features and data connectivity capabilities.
The NexSys PCS® plasma collection device receives FDA 510(k) clearance.
NexLynk DMS® software launches.
YES® Technology (plasma yield enhancing solution) for NexSys PCS® receives FDA 510(k) clearance.
TEG® 6s hemostasis analyzer receives FDA 510(k) clearance for use in U.S. adult trauma settings and begins shipment of TEG® 6s trauma cartridges.
The company moves its global headquarters from Braintree, MA, into downtown Boston.
The company acquires intellectual property assets for the TEG® 6s hemostasis analyzer system.